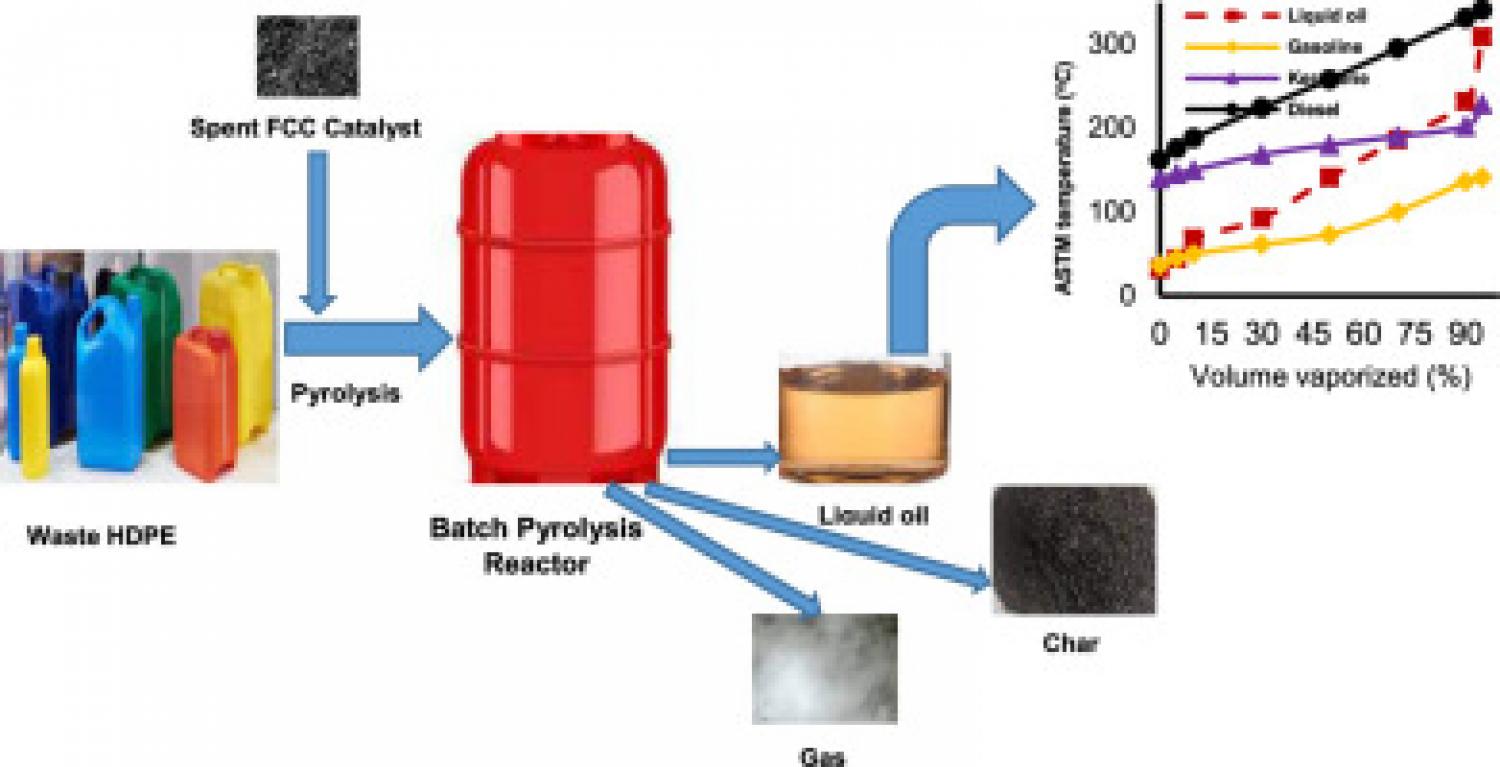
Globally expanding plastic use has created environmental issues related to the disposal of plastic waste. One of the possible alternative techniques for turning waste plastics into high-quality liquid oils is thermal and catalytic pyrolysis. The research focused on high-density polyethylene (HDPE) thermal and catalytic slow pyrolysis employing waste fluid catalytic cracking (FCC) catalyst at various temperatures and catalyst-to-plastic ratios in a batch reactor. The ASTM methods and Gas Chromatography-Mass Spectrometry (GC-MS) were used to analyze the physical and chemical characteristics of the produced liquid oil fraction. The results indicate that a pyrolysis temperature of 500°C and a catalyst to plastic ratio of 0.2 were the ideal operating conditions. The BET surface area, pore volume, and average pore size of the spent FCC catalyst were 0.103 cm2/g, 7.02 nm, and 63.24 m2/g, respectively. Waste HDPE plastic was thermally pyrolyzed, yielding 73.9 wt% liquid oil, 23.1 wt% gas, and 3 wt% char as the end products.
Furthermore, the catalytic pyrolysis produced a higher yield of liquid oil (88.8 wt%) but less gas (9.9 wt%) and char (1.3 wt%). The kinematic viscosity, density, flash point, pour point, and calorific value of liquid oil produced by catalytic pyrolysis were 2.48 cSt, 0.85 g/cm3, 34.5 °C, -6 °C, and 41.6 MJ/kg, respectively. According to the GC-MS data, the liquid oil's chemical composition contains 38 hydrocarbons between C6 and C24. Because of this, the liquid oils made from waste HDPE have characteristics similar to those of conventional fuels and can be used as alternative renewable energy sources
