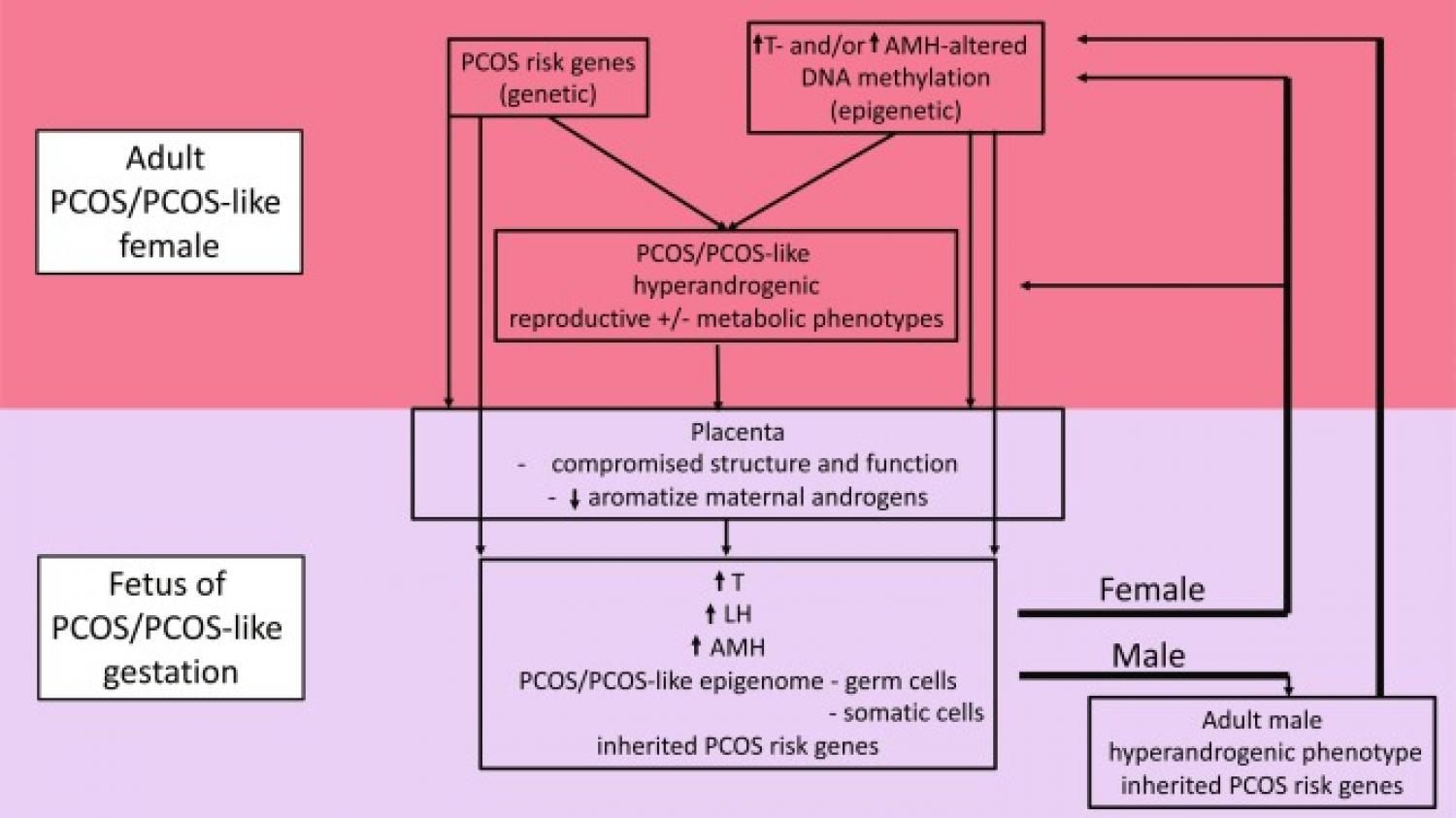
Up to 20% of women in their reproductive years have polycystic ovary syndrome (PCOS), a complex reproductive, metabolic, and behavioral women's disorder bestowing subfertility, increased obesity, and type 2 diabetes, as well as increased risk of anxiety and depression. While its origins are unknown, PCOS is highly heritable and increasing evidence suggests a combination of genetic, epigenetic, and developmental origins commencing during hyperandrogenic gestations. A reliable biomarker of female fetal hyperandrogenism, elongated anogenital distance, found in newborn daughters of women with PCOS, and in adult PCOS women, considered together with repeated confirmation of PCOS-like phenotype induction in nonhuman primate, sheep, rat, and mouse models by experimental induction of fetal female hyperandrogenism, and transgenerational transmission of induced PCOS-like phenotypes, all suggest in utero hyperandrogenic origins for PCOS pathogenesis. This brief review will focus on the evidence for PCOS pathogenesis in women and underlying molecular gateways enabling its development during hyperandrogenic gestations in animal models.
